![Figure 1: Phase diagram of carbon dioxide above and in the vicinity of the critical point showing the dense fluid, liquid-gas-like supercritical and the transitional (Widom) region. Figure 2: Sensitivity of isobaric heat capacity, cp is examined for 0.5 bar change of pressure at a constant temperature of 35 °C across a range of pressures above the critical point of pure CO2 [3]. Figure 3: Three possible transcritical CO2 compression pathways from 1 bara to 150 bara.[3] Credit: Matt Taher, P.E. Figure 1: Phase diagram of carbon dioxide above and in the vicinity of the critical point showing the dense fluid, liquid-gas-like supercritical and the transitional (Widom) region. Figure 2: Sensitivity of isobaric heat capacity, cp is examined for 0.5 bar change of pressure at a constant temperature of 35 °C across a range of pressures above the critical point of pure CO2 [3]. Figure 3: Three possible transcritical CO2 compression pathways from 1 bara to 150 bara.[3] Credit: Matt Taher, P.E.](https://cryo.memberclicks.net/assets/news/Figure%201-C02.jpg) Supercritical carbon dioxide exhibits anomalous behavior in the vicinity above the critical point. The Irish physical chemist Thomas Andrews (1863) was the first who studied the supercritical behavior of carbon dioxide. He explained his observations of the fluid state above the critical point as follows: “…the surface of demarcation between the liquid and gas became fainter, lost its curvature, and at last disappeared, the tube being then filled with a fluid which, from its optical and other properties, appeared to be perfectly homogeneous [T. Andrews, Jan. 1870]”.
Supercritical carbon dioxide exhibits anomalous behavior in the vicinity above the critical point. The Irish physical chemist Thomas Andrews (1863) was the first who studied the supercritical behavior of carbon dioxide. He explained his observations of the fluid state above the critical point as follows: “…the surface of demarcation between the liquid and gas became fainter, lost its curvature, and at last disappeared, the tube being then filled with a fluid which, from its optical and other properties, appeared to be perfectly homogeneous [T. Andrews, Jan. 1870]”.
Overview-The commercial production of oxygen and nitrogen supported the industrial revolution in the late 19th century and up to the mid-20th. Oxy-acetylene cutting and welding were important for projects like the Eiffel Tower, the Panama Canal and the Central Valley Project Corporation hydropower plants. Oxygen, nitrogen and argon refined steelmaking and metal heat treatment, from Bessemer steel converters to cryogenic tempering. Food conservation has benefited from nitrogen, argon and CO2 with the controlled atmosphere conservation of produce.
Read More
 Understanding the basic principles of dilution refrigeration is essential for operating a ZPC dilution refrigerator effectively. While there are comprehensive guides available on the subject, we provide a condensed overview for quick reference. The first principle involves the cooling of helium isotopes. Helium-4 (4He), the common isotope, liquefies at 4.2 K under standard pressure. Further cooling can be achieved by pumping on it, extracting its latent heat of vaporization. However, the cooling power is limited by the exponentially decreasing vapor pressure of helium. This restricts the achievable temperature to around 0.8 K, realistically reaching 1.2 K in the laboratory.
Understanding the basic principles of dilution refrigeration is essential for operating a ZPC dilution refrigerator effectively. While there are comprehensive guides available on the subject, we provide a condensed overview for quick reference. The first principle involves the cooling of helium isotopes. Helium-4 (4He), the common isotope, liquefies at 4.2 K under standard pressure. Further cooling can be achieved by pumping on it, extracting its latent heat of vaporization. However, the cooling power is limited by the exponentially decreasing vapor pressure of helium. This restricts the achievable temperature to around 0.8 K, realistically reaching 1.2 K in the laboratory.
Read More
 Northrop Grumman Corporation’s SpaceLogistics has sold its third Mission Extension Pod (MEP), a propulsion “jet pack” that extends the life of legacy satellites, allowing them to operate longer and more profitably. Under a purchase agreement with Intelsat, SpaceLogistics’ Mission Robotic Vehicle (MRV), a servicing spacecraft equipped with robotic arms, will install the jet pack on an Intelsat communications satellite in geosynchronous Earth orbit (GEO) to extend its life by at least six years. This capability builds on a long-standing relationship between SpaceLogistics and Intelsat that includes the world’s only commercial satellite-servicing missions in GEO.
Northrop Grumman Corporation’s SpaceLogistics has sold its third Mission Extension Pod (MEP), a propulsion “jet pack” that extends the life of legacy satellites, allowing them to operate longer and more profitably. Under a purchase agreement with Intelsat, SpaceLogistics’ Mission Robotic Vehicle (MRV), a servicing spacecraft equipped with robotic arms, will install the jet pack on an Intelsat communications satellite in geosynchronous Earth orbit (GEO) to extend its life by at least six years. This capability builds on a long-standing relationship between SpaceLogistics and Intelsat that includes the world’s only commercial satellite-servicing missions in GEO.
Read More
 Swift Aircraft, the visionary company behind the innovative British aerobatic light aircraft called the Swift, has joined forces with a team of British innovators to drive sustainable flight forward. Their collaboration is supported by the Project MONET contract, awarded by the UK Ministry of Defence (MoD), aimed at demonstrating technologies for achieving net-zero emissions during flight.
Swift Aircraft, the visionary company behind the innovative British aerobatic light aircraft called the Swift, has joined forces with a team of British innovators to drive sustainable flight forward. Their collaboration is supported by the Project MONET contract, awarded by the UK Ministry of Defence (MoD), aimed at demonstrating technologies for achieving net-zero emissions during flight.
Read More
 At CERN’s Neutrino Platform on the Laboratory’s Prévessin site in France sit two large boxes encased in a red grating. Inside these boxes are vast chambers surrounded by shiny stainless steel. The boxes are the cryostat modules of the ProtoDUNE experiment. Despite their large size, they are tiny in comparison to the future size of their successors for the Deep Underground Neutrino Experiment (DUNE), a vast neutrino experiment currently being built in the USA. The Neutrino Platform also houses an assembly station for the Tokai to Kamioka (T2K) experiment, another vast neutrino facility in Japan.
At CERN’s Neutrino Platform on the Laboratory’s Prévessin site in France sit two large boxes encased in a red grating. Inside these boxes are vast chambers surrounded by shiny stainless steel. The boxes are the cryostat modules of the ProtoDUNE experiment. Despite their large size, they are tiny in comparison to the future size of their successors for the Deep Underground Neutrino Experiment (DUNE), a vast neutrino experiment currently being built in the USA. The Neutrino Platform also houses an assembly station for the Tokai to Kamioka (T2K) experiment, another vast neutrino facility in Japan.
Read More
Squeezing Superconductors
![Image: Experiments on a family of cuprate superconductors resolve discrepancies in previous work and elucidate why the critical temperature varies with pressure. Credit: A. C. Mark et al. [1] Image: Experiments on a family of cuprate superconductors resolve discrepancies in previous work and elucidate why the critical temperature varies with pressure. Credit: A. C. Mark et al. [1]](https://cryo.memberclicks.net/assets/news/Squeezing%20superconductors.png) How electrons pair in cuprate superconductors depends, in part, on the crystalline landscape the electrons occupy. That landscape can be altered by applying pressure, which in turn causes the critical temperature (Tc) in some cuprates to rise, fall, and rise again as the pressure grows. But are the changes in Tc due to the atoms being pressed into a new crystalline structure or just being squeezed closer together? Experimental evidence is contradictory. To resolve the question, Alexander Mark of the University of Illinois Chicago and his collaborators performed experiments on the three members of the BSCCO family of cuprates [1]. They showed that the pressure-induced shifts in Tc coincide with changes in how the materials compress, which in turn reflect changes in electronic structure.
How electrons pair in cuprate superconductors depends, in part, on the crystalline landscape the electrons occupy. That landscape can be altered by applying pressure, which in turn causes the critical temperature (Tc) in some cuprates to rise, fall, and rise again as the pressure grows. But are the changes in Tc due to the atoms being pressed into a new crystalline structure or just being squeezed closer together? Experimental evidence is contradictory. To resolve the question, Alexander Mark of the University of Illinois Chicago and his collaborators performed experiments on the three members of the BSCCO family of cuprates [1]. They showed that the pressure-induced shifts in Tc coincide with changes in how the materials compress, which in turn reflect changes in electronic structure.
Read More
 Aspen Aerogels, a leading technology company specializing in aerogel-based sustainability and electrification solutions, has announced the grand opening of its state-of-the-art engineering and rapid prototyping facility in Marlborough, MA. This 59,000-square-foot facility, known as the Advanced Thermal Barrier Center (ATBC), will serve as the engineering hub for PyroThin cell-to-cell barriers, which play a vital role in optimizing the safety and performance of battery packs for the rapidly growing eMobility and energy storage system (ESS) markets.
Aspen Aerogels, a leading technology company specializing in aerogel-based sustainability and electrification solutions, has announced the grand opening of its state-of-the-art engineering and rapid prototyping facility in Marlborough, MA. This 59,000-square-foot facility, known as the Advanced Thermal Barrier Center (ATBC), will serve as the engineering hub for PyroThin cell-to-cell barriers, which play a vital role in optimizing the safety and performance of battery packs for the rapidly growing eMobility and energy storage system (ESS) markets.
Read More
 Semiconductors are foundational components of modern energy, communication, and myriad other technologies. Research on tailoring the underlying nanostructure of semiconductors for optimizing device performance has been ongoing for decades. Now, in a study recently published in Scientific Reports, researchers from the University of Tsukuba and collaborating partner UNISOKU Co., LTD., have facilitated technology development—easy-to-use, time-resolved scanning tunneling microscopy (STM)—for measuring the movement of electrons in nanostructures at high temporal and spatial resolution, in a manner that will be invaluable for optimizing nanostructure performance.
Semiconductors are foundational components of modern energy, communication, and myriad other technologies. Research on tailoring the underlying nanostructure of semiconductors for optimizing device performance has been ongoing for decades. Now, in a study recently published in Scientific Reports, researchers from the University of Tsukuba and collaborating partner UNISOKU Co., LTD., have facilitated technology development—easy-to-use, time-resolved scanning tunneling microscopy (STM)—for measuring the movement of electrons in nanostructures at high temporal and spatial resolution, in a manner that will be invaluable for optimizing nanostructure performance.
Read More
 On Tuesday, May 22, NASA’s NICER (Neutron Star Interior Composition Explorer), an X-ray telescope on the International Space Station, developed a “light leak,” in which unwanted sunlight enters the instrument. While analyzing incoming data since then, the team identified an impact to daytime observations. Nighttime observations seem to be unaffected. The team suspects that at least one of the thin thermal shields on NICER’s 56 X-ray Concentrators has been damaged, allowing sunlight to reach its sensitive detectors.
On Tuesday, May 22, NASA’s NICER (Neutron Star Interior Composition Explorer), an X-ray telescope on the International Space Station, developed a “light leak,” in which unwanted sunlight enters the instrument. While analyzing incoming data since then, the team identified an impact to daytime observations. Nighttime observations seem to be unaffected. The team suspects that at least one of the thin thermal shields on NICER’s 56 X-ray Concentrators has been damaged, allowing sunlight to reach its sensitive detectors.
Read More
 Among the most fundamental questions in astronomy: how did the first stars and galaxies form? NASA’s James Webb Space Telescope is already providing new insights into this question. One of the largest programs in Webb’s first year of science is the JWST Advanced Deep Extragalactic Survey, or JADES, which will devote about 32 days of telescope time to uncover and characterize faint, distant galaxies. While the data is still coming in, JADES already has discovered hundreds of galaxies that existed when the universe was less than 600 million years old. The team also has identified galaxies sparkling with a multitude of young, hot stars.
Among the most fundamental questions in astronomy: how did the first stars and galaxies form? NASA’s James Webb Space Telescope is already providing new insights into this question. One of the largest programs in Webb’s first year of science is the JWST Advanced Deep Extragalactic Survey, or JADES, which will devote about 32 days of telescope time to uncover and characterize faint, distant galaxies. While the data is still coming in, JADES already has discovered hundreds of galaxies that existed when the universe was less than 600 million years old. The team also has identified galaxies sparkling with a multitude of young, hot stars.
Read More
Tiny Quantum Electronic Vortexes in Superconductors Can Circulate in Ways Not Seen Before
 Within superconductors there are little tornadoes of electrons known as quantum vortices that can occur, vortices that have important implications in superconducting applications such as quantum sensors. Now a new kind of superconducting vortex has been found, according to an international team of researchers.[1]
Within superconductors there are little tornadoes of electrons known as quantum vortices that can occur, vortices that have important implications in superconducting applications such as quantum sensors. Now a new kind of superconducting vortex has been found, according to an international team of researchers.[1]
Read More
Decoding Superconductivity: “Charge Density Wave” Linked to Atomic Distortions in Superconductor
 Precision measurements reveal connection between electron density and atomic arrangements in charge-ordered states of a superconducting copper-oxide material. Researchers at the Brookhaven National Laboratory have discovered a direct connection between the disappearance of certain atomic vibrations and the emergence of a “charge density wave” in superconducting copper-oxide materials. This discovery, achieved through precision measurement, uncovers a vital relationship between atomic structure and charge distribution, advancing our understanding of superconductivity.
Precision measurements reveal connection between electron density and atomic arrangements in charge-ordered states of a superconducting copper-oxide material. Researchers at the Brookhaven National Laboratory have discovered a direct connection between the disappearance of certain atomic vibrations and the emergence of a “charge density wave” in superconducting copper-oxide materials. This discovery, achieved through precision measurement, uncovers a vital relationship between atomic structure and charge distribution, advancing our understanding of superconductivity.
Read More
 Ziath, an Azenta Life Sciences company, announced a revolution in cryogenic sample management – the Ri-Track Mirage 2D barcoded tube whole rack reader. Traditionally, identifying the identity of frost-covered racks of 2D barcoded tubes taken from low temperature storage has been a time-consuming and error-prone process. Drawing upon proprietary Radio Frequency Identification (RFID) tagging technology – the Ri-Track Mirage can reliably read 2D barcoded racks covered in ice and with racks taken directly from vapor-phase liquid nitrogen storage. The need to warm your rack to thaw it out, which may degrade thermally sensitive samples, to read your identity of your sample rack is eliminated.
Ziath, an Azenta Life Sciences company, announced a revolution in cryogenic sample management – the Ri-Track Mirage 2D barcoded tube whole rack reader. Traditionally, identifying the identity of frost-covered racks of 2D barcoded tubes taken from low temperature storage has been a time-consuming and error-prone process. Drawing upon proprietary Radio Frequency Identification (RFID) tagging technology – the Ri-Track Mirage can reliably read 2D barcoded racks covered in ice and with racks taken directly from vapor-phase liquid nitrogen storage. The need to warm your rack to thaw it out, which may degrade thermally sensitive samples, to read your identity of your sample rack is eliminated.
Read More
Researchers Investigate Reliability of Cryogenic Vacuum Distillation for Soil Water Extraction
 The stable isotopes hydrogen and oxygen (δ2H and δ18O, respectively) in soil water are widely used in ecological studies, which rely on the accurate extraction of unfractionated water from different soil types. Cryogenic vacuum distillation (CVD) is the laboratory-based technique most widely used in eco-hydrological studies. However, the reliability of this technique in reflecting soil water δ2H and δ18O is still of concern.
The stable isotopes hydrogen and oxygen (δ2H and δ18O, respectively) in soil water are widely used in ecological studies, which rely on the accurate extraction of unfractionated water from different soil types. Cryogenic vacuum distillation (CVD) is the laboratory-based technique most widely used in eco-hydrological studies. However, the reliability of this technique in reflecting soil water δ2H and δ18O is still of concern.
Read More
New Approach for the Identification of Metabolites Using LC with Ion Mobility and Cryogenic Spectroscopy
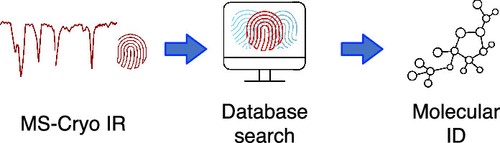 Researchers from Polytechnique Fédérale de Lausanne in Switzerland have developed a new approach for identifying isomeric and isobaric metabolites using high-resolution ion mobility spectrometry (IMS) and cryogenic infrared (IR) spectroscopy. The complex structure of metabolites has made their identification challenging, and analytical standards are often required to confirm their presence in a sample. However, the new approach can separate isomeric metabolites in a matter of milliseconds and provide highly structured IR fingerprints for their unambiguous identification. Additionally, this approach allows for the automatic identification of metabolite isomers by comparing their IR fingerprints with those in a database, thereby eliminating the need for analytical standards.
Researchers from Polytechnique Fédérale de Lausanne in Switzerland have developed a new approach for identifying isomeric and isobaric metabolites using high-resolution ion mobility spectrometry (IMS) and cryogenic infrared (IR) spectroscopy. The complex structure of metabolites has made their identification challenging, and analytical standards are often required to confirm their presence in a sample. However, the new approach can separate isomeric metabolites in a matter of milliseconds and provide highly structured IR fingerprints for their unambiguous identification. Additionally, this approach allows for the automatic identification of metabolite isomers by comparing their IR fingerprints with those in a database, thereby eliminating the need for analytical standards.
Read More
 The pressure to reduce greenhouse gas emissions continues as does the search for ‘greener energy solutions’ and one of the consequences of this is significant growth in the use of Liquid Natural Gas for marine applications. LNG has long been regarded as a sustainable fuel and has gained significant traction as a viable alternative for a wide range of commercial transport applications, including marine and shipping. It is estimated that 3% of global greenhouse gas emissions such as carbon dioxide is generated by maritime traffic, which is why the International Maritime Organization (IMO) is increasingly looking to reduce CO2 emissions. While LNG is a much ‘greener’ fuel than the highly viscous diesel fuels typically used to power container ships and cruise liners, it does present significant challenges to fuel systems and pumps, specifically dealing with extreme (cryogenic) temperatures.
The pressure to reduce greenhouse gas emissions continues as does the search for ‘greener energy solutions’ and one of the consequences of this is significant growth in the use of Liquid Natural Gas for marine applications. LNG has long been regarded as a sustainable fuel and has gained significant traction as a viable alternative for a wide range of commercial transport applications, including marine and shipping. It is estimated that 3% of global greenhouse gas emissions such as carbon dioxide is generated by maritime traffic, which is why the International Maritime Organization (IMO) is increasingly looking to reduce CO2 emissions. While LNG is a much ‘greener’ fuel than the highly viscous diesel fuels typically used to power container ships and cruise liners, it does present significant challenges to fuel systems and pumps, specifically dealing with extreme (cryogenic) temperatures.
Read More
Chart Industries, Inc. and Crane Company Successfully Test a New Liquid Hydrogen Valve
 Chart Industries, Inc. and Crane Company have successfully tested a new cryogenic valve designed and built by Crane for liquid hydrogen applications. This is the first valve launched from Crane’s CRYOFLO™ line of bellow seal vacuum jacketed globe valves, designed and tested by Crane, with final validation, review, and testing conducted by Chart. Final testing was conducted at Chart’s one-of-a-kind liquid hydrogen test facility in New Prague, MN, USA, leveraging Chart’s 60 years of experience with hydrogen equipment and the facility’s availability of the extremely cold cryogenic liquid for testing. Liquid hydrogen is over 100 degrees Fahrenheit colder than liquid nitrogen, and significantly colder than the temperature limit of most cryogenic valves.
Chart Industries, Inc. and Crane Company have successfully tested a new cryogenic valve designed and built by Crane for liquid hydrogen applications. This is the first valve launched from Crane’s CRYOFLO™ line of bellow seal vacuum jacketed globe valves, designed and tested by Crane, with final validation, review, and testing conducted by Chart. Final testing was conducted at Chart’s one-of-a-kind liquid hydrogen test facility in New Prague, MN, USA, leveraging Chart’s 60 years of experience with hydrogen equipment and the facility’s availability of the extremely cold cryogenic liquid for testing. Liquid hydrogen is over 100 degrees Fahrenheit colder than liquid nitrogen, and significantly colder than the temperature limit of most cryogenic valves.
Read More
 The Innovate UK-funded CryoCMOS Consortium, led by SureCore Ltd, reports that it has successfully created new, PDK-quality, transistor models characterized for both 4 K and 77 K cryogenic operation. SureCore is using these to develop key foundation IP to enable the design of cryo-control ASICs for use in the quantum computing space. Key to supporting this activity were the accurate cryogenic measurements undertaken by Incize of Louvain-la-Neuve, Belgium.
The Innovate UK-funded CryoCMOS Consortium, led by SureCore Ltd, reports that it has successfully created new, PDK-quality, transistor models characterized for both 4 K and 77 K cryogenic operation. SureCore is using these to develop key foundation IP to enable the design of cryo-control ASICs for use in the quantum computing space. Key to supporting this activity were the accurate cryogenic measurements undertaken by Incize of Louvain-la-Neuve, Belgium.
Read More
 Helium is a gas with properties that make it useful for many different purposes, from cooling to providing lift in airships. By understanding the basics of its cryogenic principles, we can understand how this gas works and its various uses. The aspects of cryogenic science emerged in early 19th-century experiments by Faraday and Joule. Cryogenics involves subjecting materials to extremely low temperatures, usually below -150° C.
Helium is a gas with properties that make it useful for many different purposes, from cooling to providing lift in airships. By understanding the basics of its cryogenic principles, we can understand how this gas works and its various uses. The aspects of cryogenic science emerged in early 19th-century experiments by Faraday and Joule. Cryogenics involves subjecting materials to extremely low temperatures, usually below -150° C.


